 
In the fifth century BC, Leucippus and Democritus argued that all matter was composed of small, finite. The earliest recorded discussion of the basic structure of matter comes from ancient Greek philosophers, the scientists of their day. Ive already told you, for instance about the French chemist Antoine Lavoisier who proposed the Law of Conservation of Mass which states that even if matter changes in shape or form, its mass stays the same. Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to form water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is another material that can be made from hydrogen and oxygen). Use postulates of Dalton’s atomic theory to explain the laws of definite and multiple proportions. The next major developments in atomic theory didnt come along for nearly 2,300 years. The Law of Definite Proportions applies when elements are reacted together to form the same product. 16, 1927, at the Volta celebration in Como. Law of Definite Proportions states that in a given type of chemical substance, the elements are always combined in the same proportions by mass. The content of this paper is essentially the same as that of a lecture on the present state of the quantum theory delivered on Sept. Define isotopes and give examples for several elements. Describe the three subatomic particles that compose atoms. Summarize and interpret the results of the experiments of Thomson, Millikan, and Rutherford. 2.2: Development of Modern Atomic Tehory is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. By the end of this section, you will be able to: Outline milestones in the development of modern atomic theory. 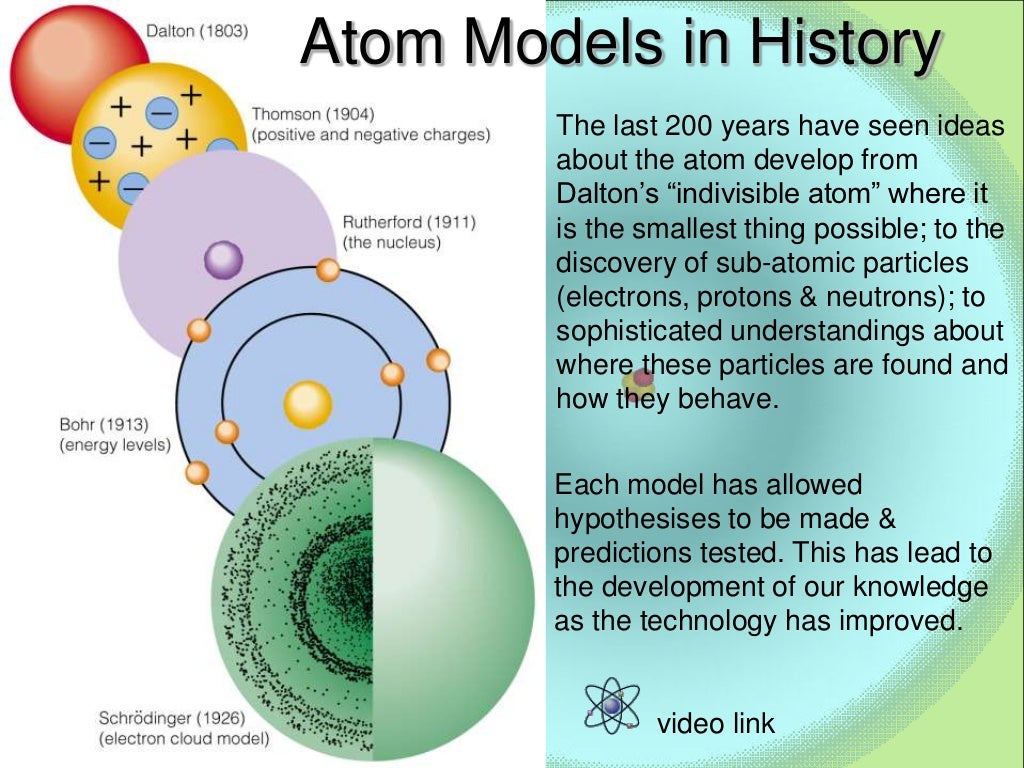
Similarly, when 2 grams of A react with 16 grams of B, they must produce 18 grams of C. Three important kinds of radiation are particles (helium nuclei), particles (electrons traveling at high speed), and rays (similar to x-rays but higher in energy). These masses are in ratio 2.66:1.33 2:1, a simple whole number ratio. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. In this equation, h is a constant known as Plancks constant, which is equal to 6. GCSE OCR Gateway Atomic structure - OCR Gateway Developing models of atoms. Second, he assumed that the energy of a photon is proportional to its frequency. Dalton drew upon the Ancient Greek idea of atoms (the word ‘atom’ comes from the Greek ‘atomos’ meaning indivisible). The mass of oxygen which combines with 1 gram of carbon to form the second oxide is 2.66. First, he assumed that light was composed of photons, which are small, discrete bundles of energy. Atoms are the smallest units of matter, and they combine to form compounds. This video is meant to provide an introduction and overview of atomic. The mass of oxygen which combines with 1 gram of carbon to form the first oxide is 1.33 grams. Take this fun History of the atomic theory quiz now and expand your knowledge. Key works through thousands of years of atomic theory, from Democritus to Chadwick. 
If 1 gram of A reacts with 8 grams of B, then by the Law of Conservation of Mass, they must produce 9 grams of C. Take a fixed given mass of carbon (element B), say 1 gram. He drew on the ideas of the Ancient Greeks in. It wasn’t until 1803 that the English chemist John Dalton started to develop a more scientific definition of the atom. It was a long wait, however, before these foundations were built upon. \)) More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles.\): If 1 gram of A reacts with 8 grams of B, then by the Law of Definite Proportions, 2 grams of A must react with 16 grams of B. Though we now know that this is not the case, their ideas laid the foundations for future atomic models. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
